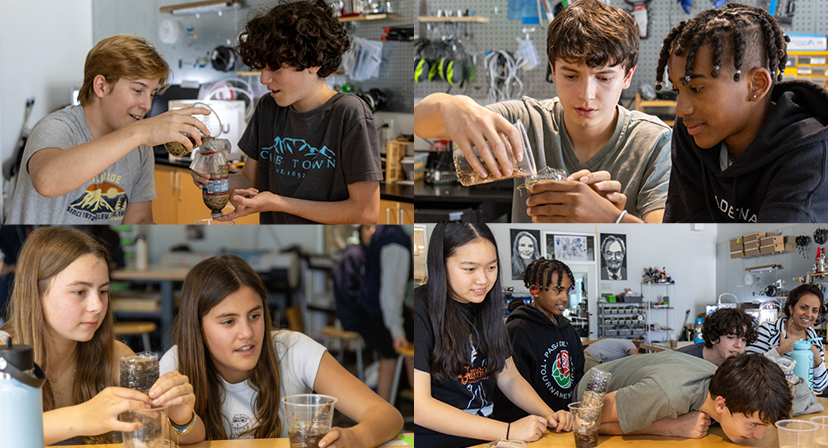Freeze Frame! Liquid Nitrogen STEM Activity: Clairbourn Kindergarteners Study Melting
 Have you ever heard a flower crackle like broken glass as it is frozen solid and shattered into tiny pieces, or seen a soft, squishy orange segment broken into hundreds of pulpy-pieces after going into a quick deep-freeze? How about a balloon filled with warm air reduced to a cracked and shriveled shell in seconds? Have you ever enjoyed ice-cream made in moments or created a dramatic fog-effect? Kindergarteners at Clairbourn had all these experiences in a hands-on lab designed to spark their interest science and the natural world.
Have you ever heard a flower crackle like broken glass as it is frozen solid and shattered into tiny pieces, or seen a soft, squishy orange segment broken into hundreds of pulpy-pieces after going into a quick deep-freeze? How about a balloon filled with warm air reduced to a cracked and shriveled shell in seconds? Have you ever enjoyed ice-cream made in moments or created a dramatic fog-effect? Kindergarteners at Clairbourn had all these experiences in a hands-on lab designed to spark their interest science and the natural world.
 The awesome substance capable of causing these cooling effects is liquid nitrogen–basically liquid distilled air that can cause rapid freezing of a variety of substances and has a wide-range of scientific uses.
The awesome substance capable of causing these cooling effects is liquid nitrogen–basically liquid distilled air that can cause rapid freezing of a variety of substances and has a wide-range of scientific uses.
Students in Mrs. Shoemake’s Kindergarten class at Clairbourn have been studying the changing properties of matter–especially melting! Previously, the class observed ice melting and predicted how long it would take for it to return completely to its liquid state. Then the class discussed what might have caused the ice to melt. Student-directed experiments followed that led them to the conclusion that heat causes and accelerates melting.
 To explore the topic further, students participated in a S.T.E.M. lab activity where orange segments, balloons, and ice-cream ingredients were frozen solid in liquid nitrogen and then brought back up to room temperature. This carefully supervised activity was organized by parent volunteers and scientists Mr. and Mrs. Buser who brought with them the necessary safety equipment such as gloves and goggles in addition to the liquid nitrogen container.
To explore the topic further, students participated in a S.T.E.M. lab activity where orange segments, balloons, and ice-cream ingredients were frozen solid in liquid nitrogen and then brought back up to room temperature. This carefully supervised activity was organized by parent volunteers and scientists Mr. and Mrs. Buser who brought with them the necessary safety equipment such as gloves and goggles in addition to the liquid nitrogen container.
 One-by-one, they helped each child place an orange segment into the liquid where it quickly frothed and boiled its way to a frozen-solid state. Once the orange slices were removed from the liquid, they were placed in a Dixie cup for the students hold as they defrosted. At that point, the orange segments were safe for the children to touch.
One-by-one, they helped each child place an orange segment into the liquid where it quickly frothed and boiled its way to a frozen-solid state. Once the orange slices were removed from the liquid, they were placed in a Dixie cup for the students hold as they defrosted. At that point, the orange segments were safe for the children to touch.
During the demonstrations, students were especially impressed by the rapid boiling of the liquid nitrogen, the gas fog produced, and the super-cool temperature of the orange slices after submersion. They were also amazed to see a balloon shrink rapidly in size when it was placed above the liquid nitrogen and then gasped when it flattened out inside the liquid, turned brittle, and began to crack!
 The final activities were using the liquid nitrogen to freeze a flower, create fog-effects, and then make ice cream in seconds with milk, cream, and sugar. The students didn’t eat the ice cream they made because they weren’t working with culinary-grade liquid nitrogen. Instead, the class enjoyed ice cream cups the Busers bought from the store.
The final activities were using the liquid nitrogen to freeze a flower, create fog-effects, and then make ice cream in seconds with milk, cream, and sugar. The students didn’t eat the ice cream they made because they weren’t working with culinary-grade liquid nitrogen. Instead, the class enjoyed ice cream cups the Busers bought from the store.
To enjoy a slide show of this activity, click here.
Clairbourn School, 8400 Huntington Drive, San Gabriel, (626) 286-3108 or visit www.clairbourn.org.